Age-related macular degeneration (AMD) affects 26% of Europeans over the age of 60, but because its early signs are not easily detected, many patients only learn that they have it after their vision has already deteriorated. A team of researchers at the Swiss Federal Institute of Technology at Lausanne (EPFL) in Switzerland has now invented a technique that makes it possible to observe the retinal cells involved in the disease in vivo. These observations, which take just a few seconds to perform, could help clinicians diagnose AMD earlier and bring about better follow-up treatment for patients, said team leader Christophe Moser, PhD.
According to the World Health Organization, 80% of the vision impairment linked to AMD could be avoided if the condition were diagnosed and treated early enough, noted study lead author Timothé Laforest, PhD. Unfortunately, standard ophthalmic exams based on visual acuity tests -- the familiar wall charts -- are not accurate enough. Even advanced imaging tools like optical coherence tomography (OCT) cannot provide a detailed view of the retina, which is a complex tissue composed of many layers, including photoreceptor segments, the retinal pigment epithelium (RPE), and the nerve fiber layer.
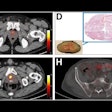
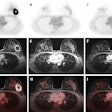
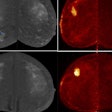
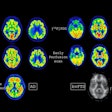
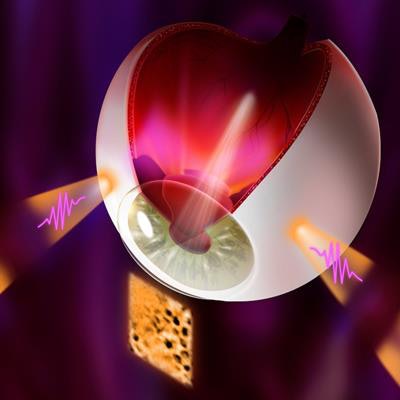
 Illuminating retinal layers provided as transscleral illumination. Image courtesy of Timothé Laforest.
Illuminating retinal layers provided as transscleral illumination. Image courtesy of Timothé Laforest.Imaging individual retinal cells within this complex tissue is difficult. Eye-motion artifacts, ocular aberrations (which reduce the lateral resolution), and the fact that retinal cells are transparent (and so have low contrast compared with surrounding cells) all contribute to the challenge. OCT, in particular, is limited because it involves sending a beam of light through the pupil. Most of this light is either absorbed or reflected at the interface between photoreceptor segments, meaning that the comparatively weak signal of light backscattered from the RPE – the layer researchers would like to study in AMD – gets overwhelmed.
"The result is that these techniques cannot provide a detailed view of the minute changes occurring in the early stages of AMD," Laforest said. "Patients are thus diagnosed when significant damage to the retina has already occurred."
Radically different method
The EPFL team developed a radically different technique for imaging retinal cells with high contrast and high resolution. Rather than sending light through the pupil, the new technique, which they have dubbed transscleral optical phase imaging (TOPI), illuminates the white part of the eye (the sclera). This tactic greatly increases the contrast of many retinal structures, as team member Mathieu Künzi explained.
One reason for the increased contrast is that although photoreceptor cells strongly reflect light that enters through the center of the pupil, their reflectivity reduces sharply for light entering via the pupil's edge. This angular-dependent reflection of the retina is known as the optical Stiles-Crawford effect, and it ensures that very little high-angle transscleral light couples to the photoreceptor cells -- meaning that a large fraction of this light instead reaches the crucial RPE layer.
Another advantage is that the angle of the light in transscleral illumination is much larger than would be possible if the light were sent in through the pupil. This high angle means that different retinal layers are not excited in a uniform way, enhancing the contrast of these transparent structures.
A third advantage stems from the fact that the team image the reflected light from the retina using a conventional transpupillary advanced optics (AO) camera system. This system corrects eye aberrations in real time, enabling them to obtain cellular-level resolution images. Importantly, it also avoids collecting any directly backscattered light because such light does not overlap with the light collection path passing through the pupil. Instead, only light that is multiply scattered by the different retina layers enters the optical system and reaches the camera.
Detecting AMD's first signs
"By imaging the RPE layer in this way, we can extract morphological parameters such as cell density and area," explained Moser. "According to ophthalmologists, this information is crucial for detecting the first signs of AMD, monitoring patients, and investigating the efficacy of new treatments."
The researchers tested their technique on ex vivo retinas of humans, rats, and pigs before obtaining approval from the Canton de Vaud Ethical Commission to apply it to 11 healthy human subjects. The results of these last-stage proof-of-concept experiments show that TOPI can quantify retinal cells from the RPE depth down to the nerve fiber layer in just 19 seconds.
"Being so fast makes TOPI compliant with routine ophthalmologic examinations in hospitals or doctors' surgeries," Laforest and Künzi said.
The researchers will soon begin a larger-scale clinical trial at the Jules Gonin Eye Hospital in Lausanne.
"The goal of this first pilot study, which will involve 100 people, is to image healthy volunteers of different ages," Laforest told Physics World. "We will also image AMD patients to qualitatively and quantitatively describe how RPE cells evolve in the process of normal aging as opposed to how they degenerate in AMD."
The TOPI technique is described in full in Nature Photonics (23 March 2020).
Belle Dumé is a contributing editor for Physics World.
© IOP Publishing Limited. Republished with permission from Physics World, a website that helps scientists working in academic and industrial research stay up to date with the latest breakthroughs in physics and interdisciplinary science.