Where are we going in cancer imaging? Prof. Dr. Regina Beets-Tan, president of ECR 2022, addressed this question in a plenary lecture on 7 October at the 2021 European Society of Magnetic Resonance in Medicine and Biology (ESMRMB) virtual meeting.
"The paradigm shift is to move from imaging the organ toward imaging the tumor microenvironment," said Beets-Tan, who is chair of radiology at the Netherlands Cancer Institute in Amsterdam.
 Prof. Dr. Regina Beets-Tan from Amsterdam. Photo courtesy of ESR.
Prof. Dr. Regina Beets-Tan from Amsterdam. Photo courtesy of ESR.In the past 10 years, there has been an exponential growth in imaging, which partly explains the increase in healthcare costs, she explained. At her own facility, an increase of up to 250% in the use of hybrid and multimodal imaging has occurred since 2008. The use of so many images in modern imaging technology only makes sense if it has an impact on patient outcome -- and in MRI, it certainly has.
In rectal cancer -- Beets-Tan's own specialty -- local recurrence rates after surgery were as high as 40% before the widespread use of MRI classification of tumors based on molecular risk factors was established in the early 2000s. With the added benefit of identifying cancer in surrounding tissue, the recurrence rate has dropped to approximately 3%.
Radiology's role
Molecular-based treatment stratification in cancer is having a real clinical impact, but radiologists may wonder where they fit in this new world, she continued.
One area is in the field of immunotherapy, she said. In the coming years, the use of immunotherapy in the neoadjuvant setting will increase, more complete responders will be identified, and new targets and genetic mutations will be discovered. Yet these advances will come at the expense of a high toxicity and high costs. Estimates for immunotherapy treatment are estimated at 180,000 euros per patient, per year.
Thus, the question becomes: Who will benefit from these treatments? The goal is to determine which patients will respond before treatment.
"We need noninvasive, advanced tools that can early predict response to immunotherapy and assess complete response in patients who undergo this in the neoadjuvant setting," she said.
To that end, advances will come by combining imaging with other biomarkers, such as those used in radiogenomics, according to Beets-Tan, who won the Most Effective Radiology Educator award in the 2019 EuroMinnies. In a recently submitted study, for instance, Beets-Tan and colleagues developed a panel of mutations in genes of different tumor types that drive the development of colon cancer.
Ultimately, the integration of tissue markers, blood, imaging, and genetic markers will enable accurate prediction models of outcomes, she said. Moreover, it will take a multidisciplinary approach.
Studies have shown that the integration of magnetic resonance radiomics, morphology, and clinicopathological markers are superior at predicting breast cancer survival than any one marker on its own.
"So we will need to get out of our own silos, and the borders between our diagnostic disciplines will fade," Beets-Tan asserted.
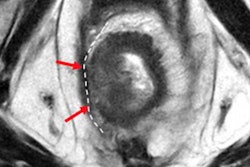
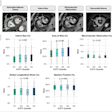
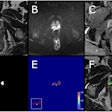
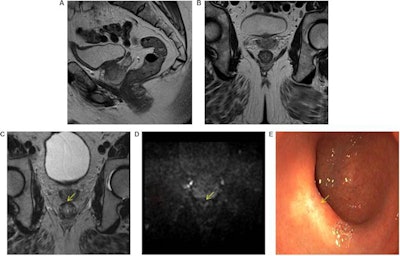 High accuracy identifying complete responders after chemoradiotherapy has been reached with the use of a three-modality approach, including digital rectal exam with endoscopy, T2-weighted MRI, and diffusion-weighted imaging. A low rectal tumor is seen on MRI (A, B). On restaging MRI eight weeks after completion of chemoradiotherapy (C), only minimal fibrosis (yellow arrow) is seen anteriorly in the rectal wall. On restaging diffusion-weighted imaging (D), there is absence of high diffusion signal (yellow arrow). Clinical assessment by endoscopy (E) reveals a white scar with telangiectasia (yellow arrow). Figure courtesy of Drs. Hester E. Haak, Monique Maas, Stefano Trebeschi, and Regina G.H. Beets-Tan and Frontiers in Oncology.
High accuracy identifying complete responders after chemoradiotherapy has been reached with the use of a three-modality approach, including digital rectal exam with endoscopy, T2-weighted MRI, and diffusion-weighted imaging. A low rectal tumor is seen on MRI (A, B). On restaging MRI eight weeks after completion of chemoradiotherapy (C), only minimal fibrosis (yellow arrow) is seen anteriorly in the rectal wall. On restaging diffusion-weighted imaging (D), there is absence of high diffusion signal (yellow arrow). Clinical assessment by endoscopy (E) reveals a white scar with telangiectasia (yellow arrow). Figure courtesy of Drs. Hester E. Haak, Monique Maas, Stefano Trebeschi, and Regina G.H. Beets-Tan and Frontiers in Oncology.Importantly, the recent decision by the European Union Commission to establish a Cancer Mission came with two key recommendations: Develop novel approaches for screening and early detection and develop a research program on early diagnostic and minimally invasive treatment technology.
"We need to work together with our clinical and diagnostic partners to search for better screening tools and prediction models," she said. "This is why I chose the theme 'Building Bridges' at ECR 2022 next year in Vienna, to provide a forum for you to meet with clinical and diagnostic disciplines and to interact and collaborate in research."