Evidence is growing that proton CT can enable more accurate treatment planning and range verification for proton therapy. Its emergence as a way to improve the precision of proton therapy was discussed at a recent forum of the European Society for Radiotherapy and Oncology (ESTRO) held in Geneva.
Proton therapy necessitates a high level of delivery accuracy in order to fully exploit the selective targeting imparted by the Bragg peak. The technique, however, comes with a number of inherent uncertainties, according to radiologist and radiation oncologist Dr. Reinhard Schulte, an associate professor in the division of physiology of Loma Linda University School of Medicine in Loma Linda, California.
 Dr. Reinhard Schulte is associate professor in the division of physiology of Loma Linda University School of Medicine in Loma Linda, California.
Dr. Reinhard Schulte is associate professor in the division of physiology of Loma Linda University School of Medicine in Loma Linda, California.
Treatment planning of passively scattered proton therapy is performed using proton ray tracing, which involves modulating the beam energy such that the weighted-echo path loss (WEPL) in tissue places the Bragg peak at the desired location. The WEPL data are derived from CT images.
"CT plays a big role in proton therapy planning for target volume delineation, and dose and range calculation," Schulte explained. "It's also used in the treatment room for 3D patient alignment and verification."
The conversion of CT Hounsfield units to proton stopping power, however, is a major source of inaccuracy, leading to systematic range errors of 3% to 5% for soft tissues and higher for bone and lung tissues or if metal artifacts are present. "Range uncertainty creates a conflict between healthy tissue sparing and the need to completely cover tumor tissue. I think that proton CT is the direct answer to this," Schulte told the assembled delegates, adding that proton CT could potentially reduce range uncertainties to below 1%. He noted that the problem of range uncertainty also exists with scanned-beam proton therapy if the range error exceeds the longitudinal width of the Bragg peak.
There's also motion uncertainties to consider, where the position of tissues relative to the treatment beam varies between, or even during, treatment fractions. In the longer term, using proton CT for image guidance could help address this problem.
Imaging with protons
Proton CT utilizes the proton beam itself to create images. The technique works by sending a beam of low-intensity protons through the patient and measuring the position, direction, and energy loss of each exiting proton. This allows reconstruction of the volumetric distribution of the relative stopping power of the protons, for use in treatment planning and pretreatment range verification.
High proton energies are required to provide sufficient range for imaging, with a 330 megaelectron volt (MeV) proton beam giving a 60-cm range, sufficient to travel right through any body part of a patient. For head scans, a lower energy of 230 MeV will suffice.
The scanner also needs to be rotatable to deliver 3D information. The first systems for performing such proton imaging are now under development. Schulte highlighted the work of the Proton CT Collaboration, a group of scientists that first met in 2003 and completed construction of their phase one (preclinical) scanner in 2010.
The phase one scanner uses two tracker modules (front and rear) to measure the location and direction of individual protons traveling through a patient. Each module has two detector boards containing two 9 x 9-cm silicon strip detectors, with the strips on each silicon strip detector oriented in either the x or y direction and a total sensitive area of 9 x 18 cm. After traversing the patient, the residual energy of each outgoing proton is measured by a segmented crystal calorimeter and used to reconstruct WEPL values.
In 2011, the team received further funding from the National Institute of Biomedical Imaging and Bioengineering and the National Science Foundation to develop a phase two scanner. This system, which should be completed by the summer of this year, offers a larger detection area of 9 x 36 cm, making it suitable for head scanning. The next-generation unit includes an improved silicon tracker based on novel silicon strip detectors with slim edges developed by researchers from the Naval Research Laboratory and the University of California at Santa Cruz. This design enables tiling of the strips with minimal dead area and no need for overlap. The crystal calorimeter is replaced by a faster multistage energy detector composed of five plastic scintillators that offers a WEPL resolution of roughly 3 mm.
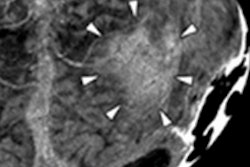
Schulte went on to present some initial results from the phase one proton CT scanner, including scans of the Lucy phantom (Standard Imaging) and a pediatric head phantom. Scans were reconstructed using an iterative algorithm (DROP-TVS) developed by former graduate student Scott Penfold from the University of Wollongong in New South Wales, Australia, and Yair Censor from the University of Haifa in Israel. The calculated relative stopping power values for acrylic, polystyrene, and bone-equivalent inserts agreed to within 1% of predicted values. Proton CT images were free of artifacts from bone.
Schulte also emphasized that, although yet to be demonstrated by measurements, proton CT delivers a head scan dose of between 0.03 and 0.3 mGy, comparable with the dose from a single chest x-ray. "The dose advantage compared with using x-ray CT for daily verification is potentially very large," he said.
He concluded by listing some applications for proton CT, including improving treatment plans in the presence of x-ray CT artifacts, and treatment planning where a very high range accuracy is required. In the longer-term, proton CT could be used for in-room pretreatment verification and adaptive therapy based on daily plan updates, as well as to enable low-dose image-guided proton therapy. Further into the future, we may even see the implementation of pretreatment proton CT and intratreatment proton radiography.
"Proton CT and proton radiography are evolving technologies driven by the need for accurate range prediction and verification," Schulte concluded. "Widespread use can be expected within the next 10 years as proton therapy expands its role in radiotherapy."
© IOP Publishing Limited. Republished with permission from medicalphysicsweb, a community website covering fundamental research and emerging technologies in medical imaging and radiation therapy.