Quibim has secured the CE mark for QP-Prostate as a class IIB device, as well as receiving UKCA certification.
The company said this allows Quibim to commercialize QP-Prostate in Europe. The device received 510(k) clearance from the U.S. Food and Drug Administration in 2021.
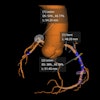
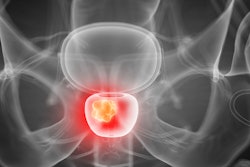
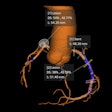
Powered by artificial intelligence, QP-Prostate is an MRI-based software aims to increase diagnostic accuracy and early detection of prostate cancer. The company said it can reduce interpretation time and help define regional diagnosis faster.