La Ciotat, France-based Avicenna.AI has received medical device regulation (MDR) certificates for five of its algorithms.
The certification addresses clinical evaluation, postmarket surveillance, device traceability, technical documentation, and risk management for products and is a critical component of maintaining their CE-mark status, which allows devices to be freely traded in Europe, the company noted.
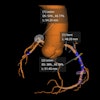
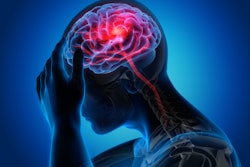
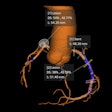
The company's MDR-certified AI tools include algorithms for intracranial hemorrhage, large vessel occlusion, quantification of stroke severity, aortic dissection, pulmonary embolism, and opportunistic cases of pulmonary embolism.